Abstract
Purpose
Unidentified dynamics for the causative agent of COVID-19, SARS-Cov-2, led to the critical public health issue. Suspicion for the airborne potential of SARS-Cov-2 is an important problem for its transmission and relevant epidemics. This research investigated hospital indoor air quality to SARS-Cov-2 occurrence and determination its air born potential.
Methods
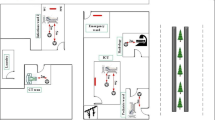
The site study was a referral hospital with 630 beds for admitting of COVID-19 patients. Air sampling was done (n = 31) on selected wards including Emergency 1, Emergency 2, bedridden (4-B, 10-D), ICU 2, ICU 3, CT-SCAN, and laundry. The average temperature and relative humidities were 22 ± 1 °C and 43 percent respectively. All glass impinger used for sampling in which the sampling pumps capacities were 5 and 40 L.min− 1. Sampling duration time was 20 and 15 minutes and 100 to 1000 L of air were gathered. All parts of the sampling equipment were completely disinfected by hot water, ethanol (70%), chlorine solution (1000 ppm), hot water (70 °C for 1 min) and washed with distilled water. The transmitting media (7 ml) was injected into impinger and residual of this media (2 ml) was sent to the virology laboratory within 2 hours and preserved on refrigerator < 4 °C. Analysis of samples was performed by RT-PCR and repeated for accuracy control.
Results
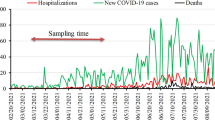
All of the samples were negative for SARS-Cov-2 occurrence. These results showed that SARS-Cov-2 had not airborne potential in this hospital.
Conclusions
Although SARS-Cov-2 similar to the SARS virus but, SARS-Cov-2 is not an airborne virus.
Similar content being viewed by others
Introduction
Induced respiratory illnesses by viral infections are the most frequent causes of morbidity and mortality worldwide [1]. SARS-CoV-2 is a member of the coronavirus family which temporarily named as SARS-Cov-2 and the causative agent of COVID-19 [2]. The COVID-19 as an acute respiratory viral disease emerged in the last days of 2019 and on March 11, 2020, WHO declared its pandemic scale [3, 4].
The dynamics of SARS-Cov-2 are now unknown, but there is speculation that it has an animal origin and have the airborne potential [2]. Besides the indefinite treatment process for COVID-19 and lack of a vaccine against its responsible pathogen, suspicion for the airborne potential of SARS-Cov-2 is another probable challenge for SARS-Cov-2 transmission and management of related pandemics [5, 6]. These problems led to inducing regional and global release potential of virus, the elevation of the basic reproduction number, R0, (Basic reproduction number) of COVID-19, occurring of several thousands of morbidity, mortality, and complication of health care services worldwide [7]. To date, over 200 developed and developing countries are suffering from COVID-19, so, in many of these countries, the health care services resiliency has critical issues for patients management [3].
Different, indistinguishable, and complicated transmission mechanisms of SARS-Cov-2 are a crucial challenge for health care policymakers which led to their astonishment on epidemics control [8, 9].
Although transmission of SARS-Cov-2 was not identified completely, several transmission mechanisms including emission by respiratory droplets, direct contact with contaminated surfaces, and airborne mechanisms are introduced for its transmission [10]. Airborne characteristics of microorganisms including viruses can lead to elevation of infectious potential and complication of epidemics control. So, airborne viruses can represent major health and economic risks. In epidemics and pandemics, the efficacy of preventive action plans depend on transmission chain disconnection levels and identification of pathogenic mechanisms of agents, so, based on introduced transmission mechanisms for SARS-Cov-2, its aerobiology, and transmission paths of the virus with analyzing of microbiological content of air is very important [11].
A retrospective cohort study reported that airborne transmission may have played an important role in SARS transmission [11]. Due to airborne transmission of foot-and-mouth disease virus (FMDV: a picornavirus that causes vesicles in the mouth and feet of bovids, suids, ovids, caprids, and other cloven-hoofed animals, is highly infectious and a major plague of animal farming.) and Norwalk-like virus reported in previous studies, aerosols may also be responsible for the transmission of COVID-19 and other viral diseases. Since the airborne potential of SARS-Cov-2 is controversial, according to the abovementioned issues, the purpose of this study is the investigation of the airborne potential of SARS-Cov-2 in a large hospital as representative of medical staff workplace.
Materials and methods
This research was investigated by the indoor air quality of a military hospital for the presence of SARS-Cov-2. The hospital in question was a large military hospital located in District 2 of Tehran municipality with 600 beds. Air sampling was done in selected wards of this referral hospital for admitting of COVID-19 patients, including Emergency 1, Emergency 2, bedridden (4-B, 10-D), ICU 2, ICU 3, CT-SCAN (Computed tomography scan) and laundry wards. Bed distance in rooms and ICU wards was 1 meter without any separator. All investigated wards were fully occupied in which patients had an oxygen mask. Only in the ICU ward with the intubated and critical patients, without disturbance in nursing care, the patient’s beds were separated by transparent nylon drapes from the nursing station. Air pressure in hospital wards had atmospheric conditions and not measured. Central air conditioning with positive pressure was used in hospital wards. The average temperature and relative humidity were 22 ± 1 °C and 43 percent respectively. Air sampling was performed within 5 days by all-glass impinger (AGI) [11] in which sampling pumps capacities were 5 L.min− 1 (SKS pump, P1) and 40 L.min− 1(P2). Sampling pumps were calibrated by dry air calibrator (DRY GAS METER. SHINAGAWA SEIJI CI. LTD. TOKYO, KYOTO, FUKUOKA.). Sampling duration time was 20 and 15 minutes and 100 to 1000 L of air were gathered by P1 in randomized stations and P2, respectively. Sampling with P1 was performed on randomized stations and P2 with circulation patterns and without stand stations. Air sampling setup installed about 1.2 to 1.8 m from the floor and approximately 0.5 to 4 m away from patients’ beds. Before each sampling, all parts of the sampling equipment including AGI parts and vessels completely disinfected by hot water (70 °C for 1 min), chlorine solution (1000 ppm), ethanol (70%), hot water (70 °C for 1 min) and washed with sterile distilled water. The volume of air collected was in the range of 100 to 1000 L. The transmitting media (7 ml) was injected into glass impinger and after sampling residual of this media (2 ml) were sent to virology laboratory within 2 hours. All samples were stored at 4 °C until sent to the virology laboratory. Analysis of samples was performed by RT-PCR (Reverse transcriptase polymerase chain reaction) and repeated for accuracy control. The one-step rRT-PCR (Real time Reverse transcriptase polymerase chain reaction) assay was developed using the QuantiFast RT-PCR Kit (Qiagen, Hilden, Germany), using the Corbet (Rotor-Gene) 6000 (Qiagen, Germany) and Applied Biosystems (ABI) 7500 (USA) real-time PCR instruments. The amplifications were accomplished within 1 hour, in 20 µL reaction mixtures containing 5 µL of the targeted viral RNA and 2 µL (20 pmol) of each primer (10 µM). The cycling conditions were as follows: a single cycle of 10 minutes at 50 °C and 3 minutes at 95 °C, followed by 40 cycles of 10 seconds at 95 °C, and a final cycle of 30 seconds at 60 °C [12]. The rRT-PCR products were detected via an increase in fluorescence from cycle to cycle, and the positive and negative controls were used as the quality controls for the process. The real-time analysis was evaluated with negative and positive controls. The rRT-PCR amplicons were then confirmed via electrophoresis.
Results and discussion
All of the patients had a severe form of cough and sneezing. Patients’ O2 saturation ranging from 54 to 93 percent. All of the patients had an oxygen mask in bedridden wards and ICUs. In ICU3 and ICU2, three and two patients were intubated, respectively. Gender classification of patients was not considered. The average of patients old in bedridden wards and ICUs were 55 and 65 years, respectively.
All of the samples were negative for SARS-Cov-2 occurrence (Table 1). Complementary information of investigated wards was informed in Table 2, which implies that all investigated wards had central, mechanical, and positive pressure ventilation systems. These results showed that SARS-Cov-2 had not airborne potential in this hospital. These results concur with WHO reports which considered in previous research by Faridi et al. [13]. Also, Faridi and et al. were investigated the airborne potential of SARS-COV-2 and reported this virus had not airborne potential [13]. Although previous research conducted using laboratory nebulizer was reported that SARS-COV-2 had aerosol transmission potential and its airborne potential is plausible [14], but this field research confirms that SARS-Cov-2 had not airborne potential. We claim that the natural mechanism of patients’ cough and sneezing are differing than experimental nebulizers which considered in previous research for the airborne potential of viruses. Harvard medical school researchers reported that clear evidence of person-to-person airborne transmission of SARS-Cov-2 has not been reported. These researchers reported that the airborne component of transmission for SARS-Cov-2 is likely based on other respiratory viruses such as SARS, MERS, and influenza and air sampling for SARS-Cov-2, in a clinical setting, has demonstrated detectable viral RNA, the extent of transmission resulting from airborne particles relative to large respiratory droplets, directly and on surfaces, is not yet known [15].
CDC was reported that natural and mechanical ventilation is effective methods for SARS-Cov-2 control. This claim concurred with the findings of the present research in which the selected wards of the hospital had a mechanical air conditioning system and negative detectable viral RNA for SARS-Cov-2 in air samples. So, it can be concluded that the natural and mechanical air conditioning system with positive pressure is an available technology for air cleaning in patients’ convents and hospitals. Also, based on Klompas et al., reports no perfect experimental data are proving or disproving droplet vs. aerosol-based transmission of SARS-CoV-2 [16]. Finally, the balanced evidence, disproving aerosol-based transmission of SARS-CoV-2 particularly in well-ventilated spaces and suggests that long-range aerosol-based transmission is not the dominant mechanism of SARS-CoV-2 transmission. All of these relevant research concurs with the results of the present study.
Conclusions
In this comprehensive research 31 samples were analyzed for the occurrence of SARS-Cov-2 as the causative agent of COVID-19 in the indoor air of a referral hospital for admitting COVID-19 patients. Although the investigated hospital had the central and positive mechanical air condition system, this research revealed that SARS-Cov-2 had not airborne potential. Mechanical air conditioning and natural ventilation are available technologies that can be used for air cleaning in patients’ convents and hospitals. Despite these results, since medical staff have close contact with patients and work in a hazardous area, the protection of medical staff should not be violated and must be considered based on national and international strict guidelines.
Abbreviations
- COVID-19:
-
Coronavirus disease of 2019
- SARS-Cov-2:
-
2019 Novel Coronavirus
- FMDV:
-
foot-and-mouth disease virus
- ICU:
-
Intensive care unit
- CCU:
-
Central care unit
- RT-PCR:
-
Reverse transcriptase polymerase chain reaction
- rRT-PCR:
-
Real time Reverse transcriptase polymerase chain reaction
- R0 :
-
Basic reproduction number
- RH:
-
Relative humidity
- CT-SCAN:
-
Computed tomography scan
References
Moattari A, Aleyasin S, Emami A, Fyruzi M, Pirbonyeh N. The prevalence of human metapneumovirus and respiratory syncytial virus and coinfection with both in hospitalized children with acute respiratory infection in South of Iran. Arch Pediatr Infect Dis. 2015;3(3):e21581.
Jiang S, Du L, Shi Z. An emerging coronavirus causing pneumonia outbreak in Wuhan, China: calling for developing therapeutic and prophylactic strategies. Emerg Microbes Infect. 2020;9(1):275–7.
Cascella M, Rajnik M, Cuomo A, Dulebohn SC, Napoli RD, Review. Features, evaluation and treatment coronavirus (COVID-19). StatPearls. StatPearls Publishing LLC, Treasure Island; 2020.
Zu ZY, Jiang MD, Xu PP, Chen W, Ni QQ, Lu GM, Zhang J. Coronavirus disease 2019 (COVID-19): A perspective from China. Radiology. 2020;200490.
Geller C, Varbanov M, Duval RE. Human coronaviruses: insights into environmental resistance and its influence on the development of new antiseptic strategies. Viruses. 2012;4(11):3044–68.
Fehr AR, Perlman S. Coronaviruses: an overview of their replication and pathogenesis. Methods Mol Biol. 2015;1282:1–23.
Zhao S, Lin Q, Ran J, Musa SS, Yang G, Wang W, et al. Preliminary estimation of the basic reproduction number of novel coronavirus (2019-nCoV) in China, from 2019 to 2020: A data-driven analysis in the early phase of the outbreak. Int J Infect Dis. 2020;92:214–7.
Khan S, Siddique R, Ali A, Xue M, Nabi G. Novel coronavirus, poor quarantine, and the risk of pandemic. J Hosp Infect. 2020.
Ong SWX, Tan YK, Chia PY, Lee TH, Ng OT, Wong M, Air, surface environmental, and personal protective equipment contamination by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) From a symptomatic patient. JAMA. 2020;323(16). https://doi.org/10.1001/jama.2020.3227.
Lai CC, Shih TP, Ko WC, Tang HJ, Hsueh PR. Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) and coronavirus disease-2019 (COVID-19): The epidemic and the challenges. Int J Antimicrob Agents. 2020;55(3):105924.
Verreault D, Moineau S, Caroline D. Methods for sampling of airborne viruses. Microbiol Mol Biol Rev. 2008;72(3):413–44.
Corman VM, Landt O, Kaiser M, Molenkamp R, Meijer A, Chu DKW, et al. Detection of 2019 novel coronavirus (2019-nCoV) by real-time RT-PCR. Euro Surveill. 2020;25(3). https://doi.org/10.2807/1560-7917.ES.2020.25.3.2000045.
Faridi S, Kaveh Sadeghi SN, Naddafi K, Yavarian J, Shamsipour M, Shafiei Jandaghi NZ, et al. A field indoor air measurement of SARS-CoV-2 in the patient rooms of the largest hospital in Iran. Sci Total Environ. 2020;725:138401.
Doremalen N, Bushmaker T, Morris DH, Phill M, Holbrook MG, Gamble A, et al. Aerosol and surface stability of SARS-CoV-2 as compared with SARS-CoV-1. N Engl J Med. 2020;382;(16):1–4.
Nardell EA, Nathavitharana RR. Airborne spread of SARS-CoV-2 and a potential role for air disinfection. JAMA. 2020;324(2):141–2. https://doi.org/10.1001/jama.2020.7603.
Klompas M, Baker MA, Rhee C. Airborne transmission of SARS-CoV-2: Theoretical considerations and available evidence. JAMA. 2020;342(5):441–2. https://doi.org/10.1001/jama.2020.12458.
Acknowledgements
Thanks to guidance and advice from “Clinical Research Development Unit of Baqiyatallah Hospital”.
Submission declaration and verification
The work has not been published previously in the form of an abstract or published lecture.
Funding
The authors declare that this study had financial support for virology tests, provided by the vice president for research of Baqiyatallah University of Medical Sciences, grant number REC. 1399.031 and research code 99000059.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
This research was performed based on Baqiyatallah University of Medical Sciences ethics committee approval as IR.BMSU.REC.1399.031.
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Masoumbeigi, H., Ghanizadeh, G., Yousefi Arfaei, R. et al. Investigation of hospital indoor air quality for the presence of SARS-Cov-2. J Environ Health Sci Engineer 18, 1259–1263 (2020). https://doi.org/10.1007/s40201-020-00543-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40201-020-00543-3